SMART-NAFLD
Systems Medicine Approach to Early Detection and Prevention of Hepatocellular Carcinoma in Non-Alcoholic Fatty Liver Disease
This consortium brings together 12 groups that are researching on metabolic-associated steatotic liver disease (MASLD, formerly known as non-alcoholic fatty liver disease (NAFLD)). The researchers characterize molecular changes in MASLD to find urgently needed innovative strategies for the identification of patients at risk to develop liver cancer.
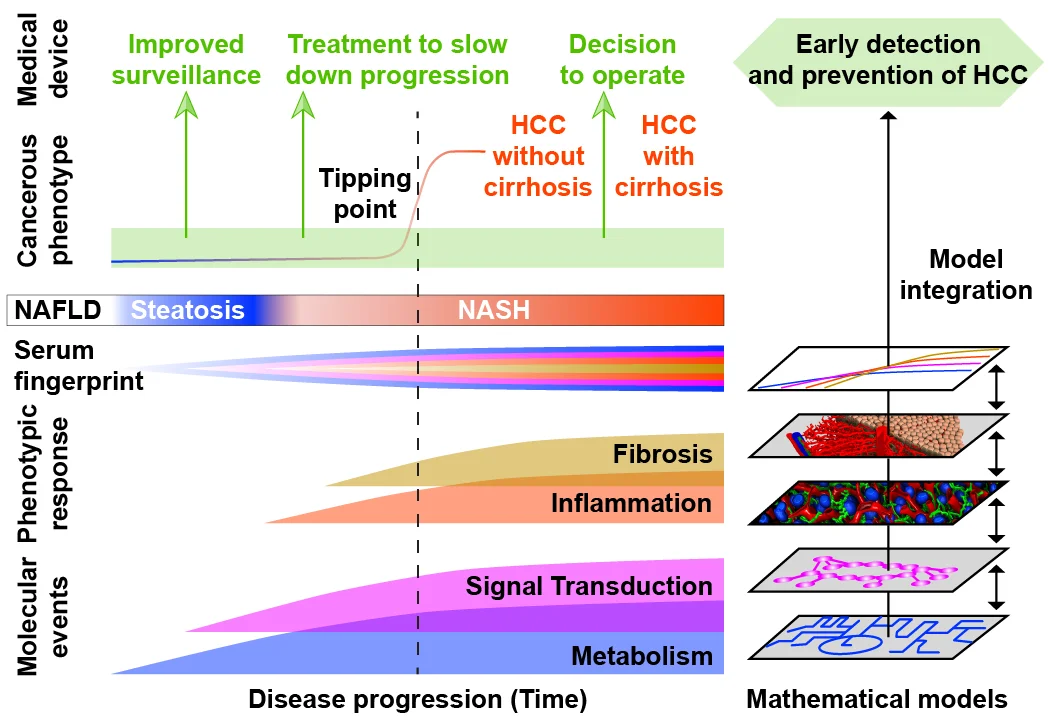
A central hypothesis in SMART-NAFLD is that altered sugar and lipid metabolism in hepatocytes of MASLD patients promotes increased cell proliferation. Together with elevated levels of proinflammatory cytokines, this metabolic state drives passing through the tipping point and thus the direct transition from MASLD to HCC formation in the non-cirrhotic liver. For the early detection of HCC, it is therefore essential to characterise tumor-promoting changes in the interaction of metabolism and signal transduction, which is induced by growth factors and / or proinflammatory factors.
To understand these complex relationships, an integrative mathematical model of metabolism and signal transduction will be developed that quantitatively describes the metabolic state and the extent of cellular reactions in hepatocytes of different stages of the disease. The integration of the cellular model into a tissue model that describes the structural changes during the course of the disease allows quantitative predictions regarding the temporal course of the disease.
Global studies of the plasma proteome and metabolome are used to identify characteristic changes in longitudinal blood samples and link them to mechanistic changes in tissue. Based on these findings a classification tool will be developed that allows early detection of MASLD patients at high risk of progression to HCC. The model is intended to support individualized therapeutic decisions for curative chirurgic therapy of early lesions.
The main objectives in SMART-NAFLD are:
- Identification of tumor-promoting changes in metabolism and signal transduction in MASLD.
- Comparison of MASLD-associated HCC with and without cirrhosis
- Linking changes in cellular responses to structural manifestations in tissue and defining indicators in the blood marking progression from MASLD to HCC
- Elucidate the gender-specific molecular and structural differences that affect the dynamics of HCC formation in MASLD.
