Semi-automated isolation of parenchymal and non-parenchymal liver cells from mice and humans with enhanced stellate cell fraction
Abstract
Background & aims
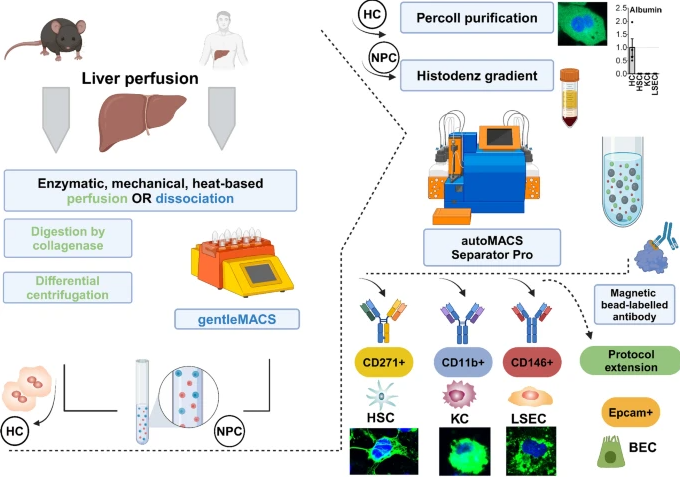
Understanding of the dynamic changes of hepatocytes, hepatic stellate cells (HSC), Kupffer cells (KC), and liver sinusoidal endothelial cells (LSEC) is critical for studying liver (patho)physiology studies. Current isolation methods often focus on single cell types, limiting utility in comprehensive research. We aiming to develop a novel, semi-automated protocol for the simultaneous isolation of hepatocytes and non-parenchymal cells (NPCs), including HSC, KC, and LSEC, from mouse and human, with high yield, purity, and viability.
Methods
The protocol employs a two-step EGTA and collagenase II perfusion for tissue digestion. Hepatocytes were isolated by low-speed centrifugation and a Percoll gradient. Subsequently, magnetic-activated cell separation, using CD271 as a selective surface marker for HSC, CD11b for KC and CD146 for LSEC) was performed. Validation was achieved with immunofluorescence staining, flow cytometry, RT-PCR, and UV fluorescence, whereby yield, purity, and viability were assessed.
Results
With our method, yield of hepatocytes, HSC, KC, and LSEC, is 33.4 ± 5.5 × 106, 5.2 ± 6.3 × 104, 12.4 ± 4.8 × 105 and 18.2 ± 8.9 × 105 cells per healthy mouse liver, respectively, with cell viabilities exceeding 89%, and purity surpassing 90%. CD271 was validated as an effective marker for purifying HSC in healthy and diseased human (n = 4–6) and mouse livers. Compared to microfluidic and organ-on-a-chip approaches, with our protocol, we achieve higher yield and purity values while enabling the simultaneous isolation of multiple cell types from a single sample.
Conclusions
Our semi-automated protocol offers a scalable, reliable, and versatile solution for isolating main liver cell types with high yield, purity, and viability from both healthy and diseased tissues, advancing liver research and facilitating downstream investigations.
